When exploring the vast catalog of engineering design materials, one of the most frequent questions posed by developers and consumers alike is: is nylon a type of plastic?
The short answer is an emphatic yes.
Nylon is a plastic, not rubber, classified as a synthetic thermoplastic polyamide (PA).
Beyond its fame in the textile world, it stands as a cornerstone synthetic polymer that has redefined modern manufacturing.
From high-performance nylon parts in industrial machinery to the common nylon fabric used in our apparel, this material bridges the gap between everyday consumer goods and heavy-duty engineering.
Understanding the transition of nylon from a laboratory curiosity to a global commodity requires a look at its unique properties.
Nylon is a thermoplastic material derived from petrochemicals, known for its exceptional strength, durability, and numerous advantageous properties.
Its ability to be engineered into different shapes—whether as thin nylon fibers or thick nylon billets—makes it a preferred choice for engineers worldwide.
The Historical Genesis of Nylon Polymers
The story of nylon is inseparable from the history of industrial chemistry in the United States.
Nylon was first developed by DuPont in the 1930s and commercialized in the 1940s, marking the dawn of the age of synthetic plastic.
This breakthrough was not merely a lucky accident but the result of rigorous acid catalysis and polymerization research.
The first nylon, nylon 66, was synthesized on February 28, 1935, by Wallace Hume Carothers at DuPont’s research facility.
Initially, the public saw nylon in very specific, small-scale applications.
Specifically, nylon was first used commercially in a nylon-bristled toothbrush in 1938 and later in women’s stockings, which became popular after their introduction at the 1939 New York World’s Fair.
However, the global landscape soon changed.
During World War II, almost all nylon production was diverted to the military for use in parachutes and other military supplies. This shift was necessitated by the loss of silk imports, forcing a rapid acceleration in nylon production capabilities.
The development of this synthetic plastic was a massive organizational feat.
In fact, nylon’s production required collaboration between multiple departments at DuPont, including the Department of Chemical Research and the Ammonia Department.
Even its name has a curious origin; the name ‘Nylon’ was derived from a modification of the term ‘norun’ (no run) to create a unique marketing name that was not trademarked.
By the time the first nylon plant began commercial production on December 15, 1939, in Seaford, Delaware, the world was ready for a material that promised durability far beyond traditional materials.
Chemical Composition and Molecular Structure
To understand why nylon behaves differently than other plastics, one must examine its molecular structure.
As a semi crystalline synthetic polymer, its internal arrangement provides a balance of flexibility and rigidity.
Nylon’s molecular structure includes repeating amide linkages, which contribute to its exceptional mechanical strength and thermal stability.
This structure allows the polymer chain to align closely, creating strong inter-chain bonds.
The Role of Raw Materials
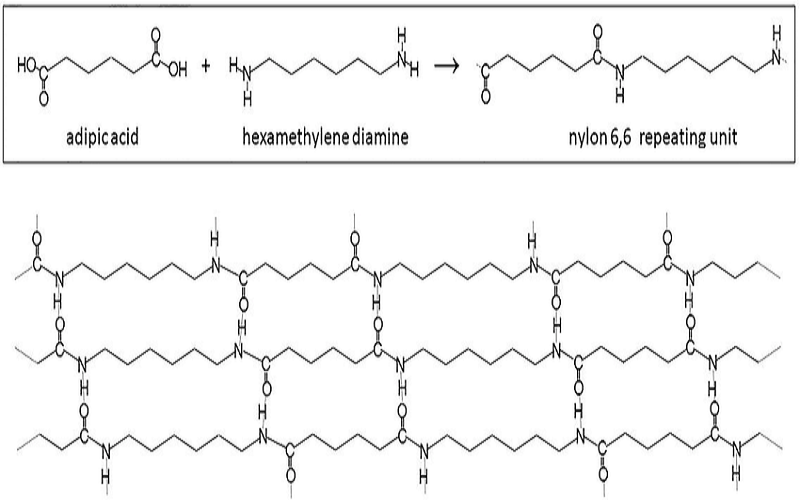
The synthesis of nylon polymers typically involves the reaction of a diamine and a dicarboxylic acid.
For instance, adipic acid and hexamethylenediamine are the raw materials for Nylon 6,6, while sebacic acid may be used for other specialized formulations.
Furthermore, nylon is characterized by its semi-crystalline structure, which provides rigidity and dimensional stability under load.
This crystallinity is why nylon is often the preferred material over brittle plastics for parts that must endure significant stress.
Thermal and Electrical Properties
In terms of its thermal properties, nylon is quite remarkable. It is classified as a thermoplastic polymer due to its ability to be melted and reshaped without significant degradation.
Additionally, its electrical properties make it a good electrical insulator with dielectric strength of 15-25 kV/mm, making it suitable for electrical applications.
Unlike common plastics, it maintains its structural integrity across a wide temperature range, though its melting point must be carefully managed during material selection.
Types of Nylon: Nylon 6 vs. Nylon 6,6
In industrial applications, not all nylons are created equal. While there are many types of nylon, two variants dominate the market.
Nylon 6 and Nylon 6,6 are the two most commonly used types of nylon in industrial applications, each offering a slightly different profile of mechanical properties and processing requirements.
Production Methods and Casting
The way these materials are formed significantly impacts their final utility.
Specifically, Nylon 6 is produced through a liquid casting process, while Nylon 6,6 is made by melting solid polymer pellets and processing them through a thermoplastic extruder.
The liquid casting process for Nylon 6 allows for the creation of large, stress-free nylon billets and rods, whereas the extrusion of Nylon 6,6 is ideal for high-speed nylon production of smaller automotive parts and nylon fabric.
Performance in Harsh Environments
When comparing the two, Nylon 6,6 generally offers higher heat resistance and lower moisture absorption than Nylon 6.
However, Nylon 6 exhibits high tensile strength, wear resistance, and low friction coefficient, distinguishing it from rubber’s elastic characteristics.
Both are engineering plastic staples, but the material selection often depends on whether the part will be exposed to high temperatures or requires specific abrasion resistance.
Mechanical Strength and Wear Propertie
Nylon is famous in the engineering world for its outstanding bearing and wear properties.
It often outperforms many metals in high-friction environments because it doesn’t require the same level of external lubrication.
Nylon’s self-lubricating properties reduce friction and wear, making it ideal for moving parts in machinery.
One of the most significant trends in industrial machinery is the replacement of heavy metal parts with nylon plastic.
Nylon is frequently used to replace metal bearings and bushings, often eliminating the need for external lubrication.
By switching to nylon, companies achieve reduced weight and better cost effectiveness.
Furthermore, nylon’s high wear resistance ensures long service life in bearing and gear applications, maintaining performance through millions of cycles without significant degradation.
To push the boundaries of nylon’s versatility, manufacturers often add reinforcing agents.
Specifically, nylon is often blended with other materials to enhance its properties, such as adding glass or carbon fiber to increase stiffness.
Carbon fiber reinforced nylon, for instance, provides high tensile strength while maintaining a low coefficient of friction.
This makes it a preferred choice for wear pads and power transmission applications where high strength is non-negotiable.

Applications of Nylon
The automotive industry is perhaps the largest consumer of nylon plastic today.
Due to its high strength and thermal stability, it is found throughout the vehicle’s architecture.
Nylon is widely used in the automotive industry for manufacturing various automotive components like engine covers, fuel tanks, and door panels.
Industrial and Automotive Applications
Modern engines run at high temperatures, requiring materials that can handle the heat without failing.
Nylon is significantly stiffer and stronger than polyethylene and has higher heat resistance, making it perfect for engine covers and intake manifolds.
In fact, nylon’s lightweight nature is a key advantage, especially when compared to traditional materials like metals, helping manufacturers meet fuel efficiency standards through reduced weight.
Consumer Goods and Packaging
Beyond the garage, nylon is found in our kitchens and stores.
Nylon is used in food packaging materials due to its moisture-resistant property and ability to seal in freshness.
Its chemical resistance also makes it popular for kitchen utensils, where it must withstand oils and heat.
Furthermore, nylon is commonly used in plastic fasteners like nuts, bolts, and washers, as well as machine components such as gears, bearings, and bushings.

Textiles and the Carpet Industry
While we often focus on nylon plastic as a rigid engineering plastic, its role in the fiber industry is massive. Nylon is the most popular fiber type in the residential carpet industry today.
Because of its high tensile strength and abrasion resistance, it can withstand years of foot traffic.
Comparison with Other Fibers
When compared to alternatives like polyester, nylon shows a unique profile. Nylon is stronger, more elastic, and softer than polyester, but absorbs more moisture and dries slower than polyester, which has better UV resistance.
This moisture absorption is a critical factor in engineering design; for instance, nylon absorbs moisture, which affects its performance, unlike polyester, which is hydrophobic.
Resilience in Textiles
The longevity of nylon stockings and carpets comes from the fiber’s molecular structure.
Specifically, nylon fibers are commonly used in the carpet industry due to their resilience, abrasion resistance, and ability to maintain their shape and color over time.
This makes nylon the preferred material for high-traffic environments where durability is paramount.
Environmental Impact and Sustainability
As we increase our reliance on synthetic plastic, the environmental impact of nylon has come under intense scrutiny.
While it is a robust polymer, its lifecycle presents significant challenges.
Discarded nylon fabric takes 30–40 years to decompose, leading to long-term landfill issues.
As we increase our reliance on synthetic plastic, the environmental impact of nylon has come under intense scrutiny.
While it is a robust polymer, its lifecycle presents significant challenges.
Discarded nylon fabric takes 30–40 years to decompose, leading to long-term landfill issues.
Recycling this engineering plastic is technically possible but economically difficult. Nylon is a robust polymer and lends itself well to recycling, but the process of recycling nylon is very expensive and difficult.
In fact, the US EPA estimates that 9.2% of carpet fiber, backing and padding was recycled in 2018, while 73% was discarded in landfills.
To mitigate this, some companies are turning to recycled nylon made from ocean waste, though the high cost remains a barrier.
The durability of nylon becomes a curse when it enters the ecosystem. Nylon used in discarded fishing gear such as fishing nets is a contributor to debris in the ocean.
Furthermore, when it is incinerated improperly, the results are dangerous.
Various nylons break down in fire and form hazardous smoke, and toxic fumes or ash, typically containing hydrogen cyanide.
Manufacturers must also account for the carbon footprint; the average greenhouse gas footprint of nylon in manufacturing carpets is estimated at 5.43 kg CO2 equivalent per kg.
Conclusion
In this comprehensive guide, we have seen that nylon is far more than just a fabric for stockings.
It is a sophisticated engineering plastic that has fundamentally changed the automotive parts and industrial machinery sectors.
Nylon’s ability to be molded into different shapes and forms makes it versatile for various manufacturing processes, from liquid casting to 3D printing with glass or carbon fiber reinforcements.
Whether it is providing chemical resistance in food packaging or offering high wear resistance in power transmission applications, nylon remains a preferred choice for its balance of high strength and reduced weight.
While we must address its environmental impact through better recycled nylon initiatives, its mechanical properties and thermal stability ensure it will remain a staple of engineering design for decades to come.

